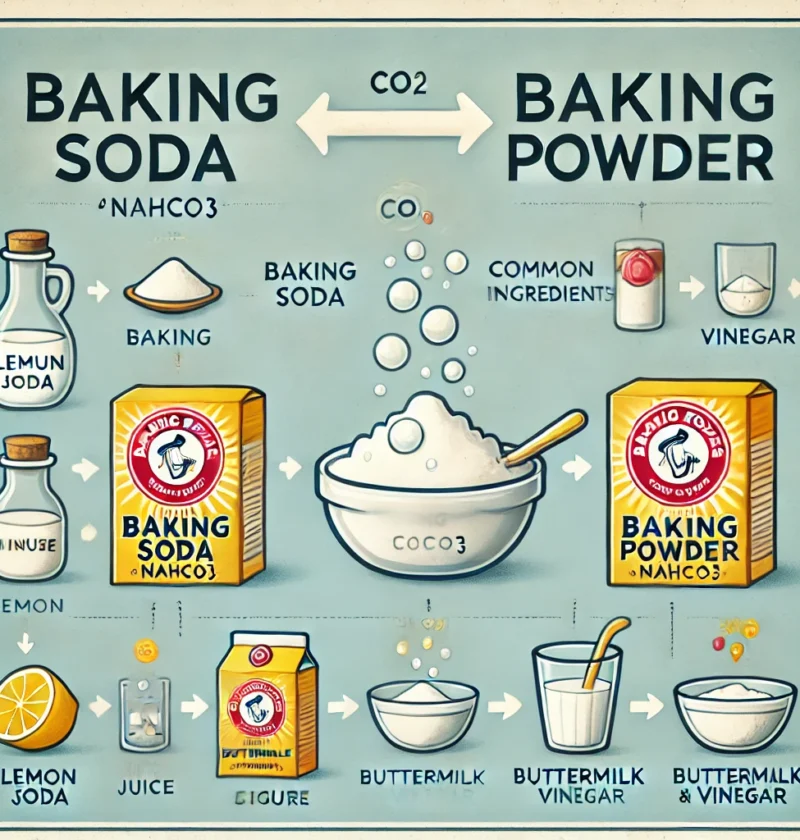
Baking soda and baking powder are two of the most commonly used ingredients in baking, yet they often cause confusion. Many home bakers wonder whether they can be used interchangeably or why a recipe calls for one instead of the other. Both are leavening agents responsible for making baked goods rise, but they work in different ways. Using the wrong one can lead to flat, dense, or overly bitter results. Understanding the difference between baking soda and baking powder is key to achieving the perfect texture and flavor in your baked treats. In this article, we’ll break down their unique roles, how they react, and when to use each for the best baking results.
What is Baking Powder?
Definition
Baking powder is a chemical leavening agent used in baking to help doughs and batters rise. It is a dry mixture of a base, an acid, and a stabilizer, which reacts to produce carbon dioxide gas when combined with moisture and heat. This gas creates air pockets in the dough, resulting in a light, airy texture in baked goods.
Chemical Composition of Baking Powder
Baking powder consists of three main components:
- Base (Alkali)
- The primary base in baking powder is sodium bicarbonate (NaHCO₃), also known as baking soda.
- When combined with an acid and liquid, sodium bicarbonate releases carbon dioxide (CO₂), causing the dough to rise.
- Acid
- The acid component is typically a weak acid that reacts with the baking soda when moistened.
- Common acids used in baking powder include:
- Monocalcium phosphate (Ca(H₂PO₄)₂) – reacts immediately when mixed with liquid.
- Sodium acid pyrophosphate (Na₂H₂P₂O₇) – reacts during baking (heat-activated).
- Sodium aluminum sulfate (NaAl(SO₄)₂) – found in double-acting baking powder, reacts at higher temperatures.
- Stabilizer (Filler or Drying Agent)
- A stabilizer is added to prevent premature activation by absorbing moisture from the air.
- Cornstarch (C₆H₁₀O₅) is the most common stabilizer, keeping the mixture dry and preventing clumping.
How Baking Powder Works
- Moisture Activation (First Reaction)
- When baking powder is mixed into the batter, the acid and base react to produce carbon dioxide gas (CO₂), forming bubbles.
- This reaction starts as soon as liquid is added.
- Heat Activation (Second Reaction in Double-Acting Baking Powder)
- If the baking powder is double-acting, a second reaction occurs when the batter is heated.
- The heat-activated acid reacts with sodium bicarbonate again, releasing more CO₂, further expanding the dough.
- This ensures a steady rise during baking, leading to a light, fluffy texture.
Types of Baking Powder
- Single-Acting Baking Powder
- Contains an acid that reacts immediately when mixed with liquid.
- Requires quick baking after mixing.
- Double-Acting Baking Powder (Most Common)
- Contains two acids—one that reacts with liquid and another that reacts with heat.
- Provides a more controlled rise, making it ideal for most baking applications.
Uses of Baking Powder
- Used in cakes, muffins, pancakes, quick breads, biscuits, and cookies to create a light, fluffy texture.
- Preferred in recipes that do not contain natural acidic ingredients (unlike baking soda, which needs an acid to activate).
- Commonly found in pre-mixed cake and pancake mixes for ease of use.
What is Baking Soda?
Definition
Baking soda, also known as sodium bicarbonate (NaHCO₃), is a basic (alkaline) compound used as a chemical leavening agent in baking. When combined with an acid and moisture, it produces carbon dioxide (CO₂) gas, which creates bubbles and causes the dough or batter to rise. It is also used for cleaning, deodorizing, and various household and industrial applications.
Chemical Composition & Properties
- Chemical formula: NaHCO₃
- Nature: Mild alkaline compound
- Solubility: Dissolves in water to form an alkaline solution
- Reaction type: Acid-base reaction
Baking soda is a weak base and remains inactive until it comes in contact with an acid.
Chemical Reactions of Baking Soda in Baking
- Basic Reaction with an Acid (Leavening Reaction)
- When baking soda reacts with an acid (like vinegar, lemon juice, buttermilk, or yogurt), it neutralizes the acid and releases carbon dioxide gas.
- This gas gets trapped in the dough, causing it to expand and rise.
- Sodium bicarbonate reacts with acetic acid to form sodium acetate, water, and carbon dioxide gas.
- Thermal Decomposition of Baking Soda (Heat Reaction) If no acid is present, baking soda can still produce CO₂ when exposed to high temperatures (above 80°C or 176°F).This reaction contributes to leavening in some baked goods, but it also leaves behind a slightly bitter, soapy taste due to sodium carbonate.
- Conclusion Baking soda is a powerful leavening agent that works best in recipes with an acidic ingredient. It produces carbon dioxide either through an acid-base reaction or heat decomposition, helping baked goods rise. However, it must be used carefully because too much can leave a bitter or soapy taste due to residual sodium carbonate.
Key Differences Between Baking Soda & Baking Powder
| Feature | Baking Soda (NaHCO₃) | Baking Powder |
|---|---|---|
| Composition | Pure sodium bicarbonate | Sodium bicarbonate + Acid + Stabilizer |
| Acid Required? | Yes, needs an external acid | No, contains its own acid |
| Reaction Type | Acid-base reaction | Self-contained reaction |
| Common Use | Recipes with acidic ingredients | General baking recipes |
| Leavening Power | Stronger but needs acid | Milder and controlled |
When to Use Baking Powder vs. Baking Soda
Use Baking Soda When:
✔ The recipe contains an acidic ingredient like:
- Vinegar
- Lemon juice
- Buttermilk
- Yogurt
- Brown sugar
- Honey
- Molasses
- Cocoa powder (natural, not Dutch-processed)
✔ You want a quick, strong reaction that starts immediately when mixed.
✔ You need browning (baking soda raises pH, aiding Maillard reaction).
✔ Example recipes: Chocolate chip cookies, pancakes, buttermilk biscuits, gingerbread.
Use Baking Powder When:
✔ The recipe does not contain an acidic ingredient to activate baking soda.
✔ You need a controlled, gradual rise (double-acting baking powder releases CO₂ twice: when mixed and when heated).
✔ You want a neutral flavor (baking soda can leave a soapy taste if not balanced with acid).
✔ Example recipes: Vanilla cakes, muffins, fluffy pancakes, scones, sugar cookies.
When to Use Both Together
✔ Some recipes use both baking soda and baking powder to balance leavening and flavor.
✔ Baking soda neutralizes acidity, while baking powder ensures consistent rising.
✔ Example: Chocolate cake (contains cocoa powder and needs extra lift).
Conclusion
Understanding the difference between baking soda and baking powder is essential for successful baking. Baking soda is a strong leavening agent that requires an acidic ingredient to activate, making it ideal for recipes with buttermilk, yogurt, or vinegar. On the other hand, baking powder contains both an acid and a base, allowing it to work independently in recipes without additional acids. While baking soda creates a quick reaction and enhances browning, baking powder provides a more controlled rise, especially in cakes and muffins. Some recipes use both to balance flavor, texture, and leavening. Choosing the right leavening agent ensures the perfect rise, texture, and taste in your baked goods.